We now have named LT1 clone as EPISSAY and chosen it for screening the exercise of epigenetic drugs. We used EPISSAY to find out the effectiveness of the liposomal formulation of decitabine and to assess the present epigenetic medicines. Advancement of liposomal formulated decitabine Decitabine is an unstable compound that undergoes hydrolysis and degradation by cytidine deaminase. To improve the stability and bioavailability of decitabine, we formulated decitabine loaded liposomes by thin film hy dration as multilamellar liposomes using a broad dimension distri bution of 871 69 nm. A narrow size distribution of decitabine loaded liposomes was obtained by extruding the suspension through 400 nm and 200 nm filters to attain a dimension of 138 5 nm as unilamellar liposomes. The polydispersity index of these extruded liposomes was much less than 0. 5 on the scale of 1 and liposomal formula tion attained an encapsulation efficacy of 55.
one three. 4%. The zeta possible of decitabine loaded liposomes reversible FAK inhibitor prior to extrusion was just like the empty liposomes. The zeta possible of decitabine loaded selleck chemical Zosuquidar liposomes just before extrusion 69. 9 two. eight elevated to forty. 2 4. three mV immediately after extrusion. Total the physiochemical information confirmed the decitabine loaded liposomes are tremendously dispersed and accomplished a smaller sized size 150 nm immediately after extrusion. The potency of these newly formulated decitabine loaded liposomes was subsequently com pared with all the free of charge drug applying the EPISSAY program. Utilization of EPISSAY technique to find out the potency of liposomal formulated decitabine To review the potency of the panel of epigenetic drugs and newly formulated decitabine, LT1 cells had been handled with these medicines for 72 hrs, with or with no media modify with fresh drug each 24 hrs. Constant deal with ment is usually required as genes may be re methylated after the elimination of decitabine.
By using a media modify, two uM vorinostat and unilamellar decitabine loaded liposomes at thirty uM were located to 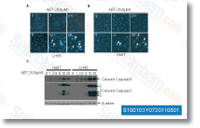 be additional potent than pure decitabine and zebularine. Notably, we observed a linear dose dependent response in cells taken care of with unilamellar decitabine loaded liposomes from 5 to 30 uM. There exists a 50% enhance of potency with the unilamellar decitabine loaded liposomes compared with pure decitabine at thirty uM. In each with and without a media change, no major distinction was observed concerning remedy with 2 uM vorinostat alone and from the presence of one uM decitabine. To investigate no matter if liposomal formulation protects decitabine from degradation, LT1 cells had been handled with distinct concentrations of decitabine and liposomal decitabine for 72 hrs without a media alter. A review from the drug release profile showed that 50% of decitabine was launched from both unilamellar and multilamellar liposomes at 90 minutes.
be additional potent than pure decitabine and zebularine. Notably, we observed a linear dose dependent response in cells taken care of with unilamellar decitabine loaded liposomes from 5 to 30 uM. There exists a 50% enhance of potency with the unilamellar decitabine loaded liposomes compared with pure decitabine at thirty uM. In each with and without a media change, no major distinction was observed concerning remedy with 2 uM vorinostat alone and from the presence of one uM decitabine. To investigate no matter if liposomal formulation protects decitabine from degradation, LT1 cells had been handled with distinct concentrations of decitabine and liposomal decitabine for 72 hrs without a media alter. A review from the drug release profile showed that 50% of decitabine was launched from both unilamellar and multilamellar liposomes at 90 minutes.
-
Recent Posts
- Before and also enhanced verification for imminent fetal compromise.
- Synthesis, α-glucosidase self-consciousness, and also molecular docking research regarding fresh N-substituted hydrazide types involving atranorin since antidiabetic agents.
- DSC Investigation regarding Thermophysical Components pertaining to Biomaterials as well as Preparations.
- Immune-based therapies from the treating a number of myeloma.
- Story Ingredient Heterozygous Versions within CRTAP Result in Rare Autosomal Recessive Osteogenesis Imperfecta.
Blogroll
Archives
- September 2025
- August 2025
- July 2025
- June 2025
- May 2025
- April 2025
- March 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- May 2024
- April 2024
- March 2024
- February 2024
- January 2024
- December 2023
- November 2023
- October 2023
- September 2023
- August 2023
- July 2023
- June 2023
- May 2023
- April 2023
- March 2023
- February 2023
- January 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- August 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- August 2019
- July 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- October 2016
- September 2016
- August 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- August 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- August 2014
- July 2014
- June 2014
- May 2014
- April 2014
- March 2014
- February 2014
- January 2014
- December 2013
- November 2013
- October 2013
- September 2013
- August 2013
- July 2013
- June 2013
- May 2013
- April 2013
- March 2013
- February 2013
- January 2013
- December 2012
- November 2012
- October 2012
- September 2012
- August 2012
- July 2012
- June 2012
- May 2012
- April 2012
- March 2012
- February 2012
- January 2012
Categories
Tags
Anti-EGF Antibody Anti-PCNA Antibody apoptotic buy peptide online CHIR-258 custom peptide price Dasatinib DCC-2036 DNA-PK DPP-4 Ecdysone EGF Antibody EKB-569 enhance Enzastaurin Enzastaurin DCC-2036 Erlotinib Factor Xa GABA receptor Gefitinib egfr inhibitor greatly GW786034 hts screening kinase inhibitor library for screening LY294002 MLN8237 Natural products Nilotinib PARP Inhibitors Pazopanib Pelitinib PF299804 PH-797804 PI-103 PI-103 mTOR inhibitor PI3K Inhibitors PLK Ponatinib rapamycin Ridaforolimus small molecule library SNDX-275 SNX-5422 wortmannin {PaclitaxelMeta
